|
Steel Making and
the Bessemer Process

|
Bessemer Process: The
Age of Iron to the Age of Steel
The
Industrial Revolution in the United States advanced with
technological advancement and invention known as the Bessemer Process.
This enabled movement from the Age of Iron to the Age of
Steel making new inventions and innovations possible
in the Second Industrial Revolution.
Steel Making and the
Bessemer Process
The Bessemer Process
was created in England by Henry Bessemer in 1855 and
brought into production by Henry Bessemer in 1860. To
create the Bessemer Process he invented a large,
pear-shaped receptacle called a converter and utilized a
blast of air in a de-carbonization process to create the
steel from iron.
Summary and
Definition of the Bessemer Process
Summary and Definition: The Bessemer
Process is the method for making steel by blasting
compressed air through molten iron to burn out excess
carbon and impurities. The Bessemer Process lowered the
cost of production steel, leading to steel being widely
substituted for cast iron. It was the creation of modern
steel.
Bessemer Process for
kids:
Brief History of Iron and Steel
Steel is an alloy of iron, with
carbon. Steel was known in antiquity and used in weapons by the
Roman Army. Steel had been produced in blast furnaces for thousands
of years and new production methods were devised in the 17th century
for blister steel and then crucible steel. Up to 3 tons of expensive
coke was burnt for each ton of blister steel that was produced.
● Wrought iron has a little
carbon (.02% to .08%), just enough to make it hard without
losing its malleability.
● Cast iron has a lot of
carbon (3% to 4.5%), which makes it hard but brittle and
non-malleable
● In between wrought and
cast iron is steel (with .2% to 1.5% carbon) making it harder
than wrought iron, yet malleable and flexible, unlike cast iron.
These properties make steel
far more useful than either wrought or cast iron but there was no
simple way to control the carbon level in iron so that steel could
be manufactured cheaply and efficiently in large quantities. In 1856
the Bessemer Process made this
possible and moved the United States into the Age of Steel.
Background History of the
Bessemer Process for kids: William Kelly
In 1847 an American
named William Kelly (1811 - 1888), the owner of an iron-works at
Eddyville, Kentucky started to experiment in processes for
converting iron into steel. William Kelly had the idea that, in the
refining process, fuel would be unnecessary after the iron was
melted if powerful blasts of air were forced into the fluid metal.
Theory behind the conversion from iron to steel was that the
heat generated by the union of the oxygen of the air with the carbon
of the metal, would accomplish the refining (burning off the
impurities). This would become known as the Bessemer Process - the
procedure to refine fluid iron by passing a stream of air through
it, which created the type of steel material used for the making of
structures.
Bessemer Process for
kids:
Henry Bessemer
The Bessemer Process
is named after the British metallurgist, engineer and inventor Sir
Henry Bessemer (1813-1898). During his career, he registered more
than 110 patents, the most famous being the converter for what would
be called the Bessemer Process. In 1877 the Royal Society of London
elected Henry Bessemer into fellowship and two years later, in 1879,
he was knighted. His involvement in steel began during the Crimean
War (1853 - 1856) when Henry Bessemer was working on the problem of
how to create more durable cannons for the British navy. Cannons
were traditionally made of cast iron but these were proving
unsuitable against new weapons which caused the cannons to explode.
Steel was the obvious choice of metal to replace cast iron but it
was expensive to produce - until he developed the Bessemer Process.
|
The Bessemer
Process: The Converter
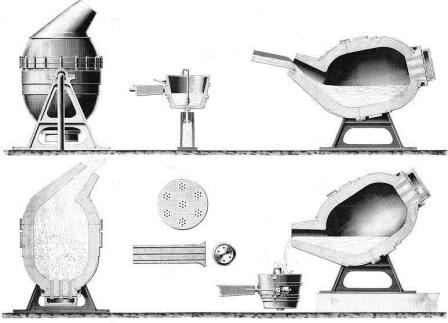
|
Bessemer Process: The
Converter
In 1856
Henry Bessemer designed what he called a converter:
● The
converter was a large, pear-shaped receptacle
● There
were holes at the bottom of the converter to allow
the injection of compressed air
● The
Bessemer converter was filled with molten pig iron
● Compressed
air was blown through the molten metal
● The
pig iron was emptied of carbon and silicon in just a
few minutes
● The
metal became even hotter and so remained molten
Bessemer Process: The
Phosphorus Problem
One problem with the early Bessemer
process was that it did not remove phosphorus from the
pig iron which makes steel excessively brittle and the
initial Bessemer process could only be used on pig iron
made from phosphorus-free ores. The phosphorus-free ores
were relatively scarce and therefore expensive.
Bessemer Process for
kids:
Sidney Gilchrist Thomas
In 1876, the Welshman Sidney
Gilchrist Thomas discovered that by adding a basic material such as
limestone to the Bessemer converter drew the phosphorus from the pig
iron into the slag. The phosphorus floated to the top of the
Bessemer converter where it could be skimmed off. This method
resulted in phosphorus-free steel - called the Basic Bessemer
process.
Significance and
Impact of the Bessemer Process
The important discovery by Sidney
Gilchrist Thomas and the improvement to the Basic
Bessemer Process was highly significant because it
meant that massive stores of iron ore from all over
the world could be used to make pig iron for
Bessemer converters. The impact of the Bessemer
process led to the inventions and innovations of the
Second Industrial Revolution. Cheap steel
revolutionized our modern world was used for the
following:
Skyscrapers ●
Railroads ●
Bridges ●
Transportation vehicles ●
Elevators ●
Weapons ●
Assembly lines ●
Kitchen Appliances ●
Surgical instruments ● Farm
implements ●
Tools, Nails and Screws ●
Tankers
Bessemer Process for kids - President Franklin Pierce Video
The article on the Bessemer Process provides an overview of one of the Important issues of his presidential term in office. The following
Franklin Pierce video will
give you additional important facts and dates about the political events experienced by the 14th American President whose presidency spanned from March 4, 1853 to March 4, 1857.
Bessemer Process
●
Interesting Facts about Bessemer Process for kids and schools
●
Definition of the Bessemer Process in US history
●
Cheap Steel Process, a Important
event in US history
●
Franklin Pierce Presidency from March 4, 1853 to March 4, 1857
●
Fast, fun, interesting
facts
about the Bessemer Process and steel
●
Foreign & Domestic
policies of President Franklin Pierce
●
Franklin Pierce Presidency and
the Significance of the Bessemer Process and steel for schools,
homework, kids and children
|